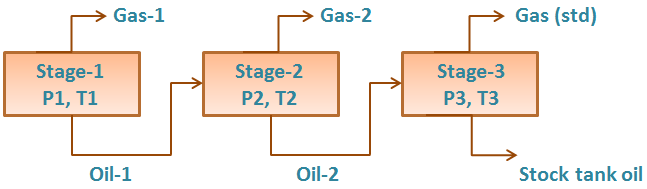
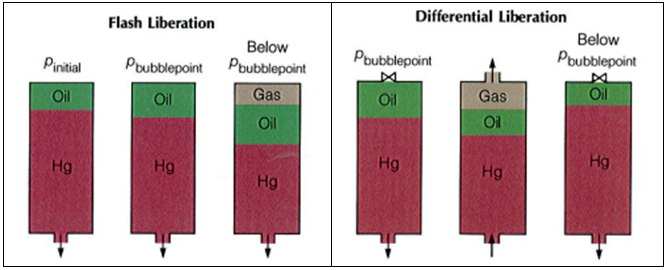
To understand the complex behavior of a reservoir fluid ” Hydrocarbon Phase Behavior “, let’s, first of all, consider the simple case of a single-compound hydrocarbon, ethane for example, initially in gas form in a cell containing mercury. As mercury is gradually injected, the gas is subjected to a continuously increasing pressure. The temperature is held constant. Actually, isothermal conditions simulate a reservoir’s generally constant temperature. At some unique pressure – the vapour pressure – the gas will condensate into a liquid.
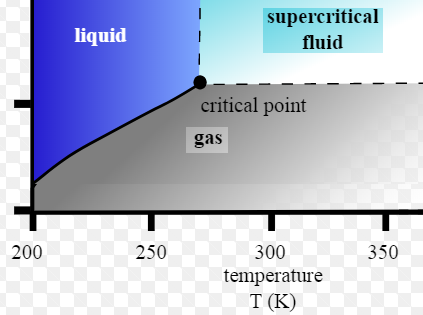
Vapour Pressure line: is the line defining the pressures at which the transition from gas to liquid occurs. Above this line, the single-compound hydrocarbon exists as a liquid, below as gas.
Critical Point: is the point at which it is no longer possible to distinguish whether the fluid is liquid or gas. The intensive properties of both phases are identical.
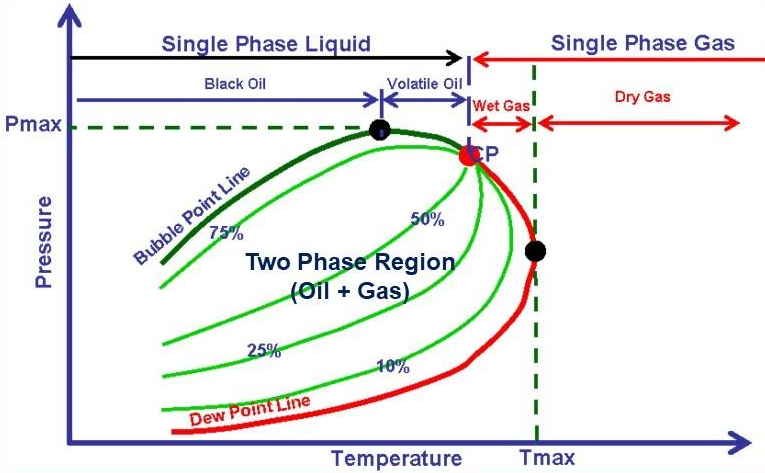
Naturally occurring hydrocarbons are more complex than the system shown above. They contain much more members of paraffin series and usually some non-hydrocarbon impurities. Nevertheless, a phase diagram can similarly be defined for complex mixtures. A typical diagram for a natural gas is shown in the following graph: